Gene Editing & Gene Therapy Development
The latest trends in gene therapy development and a brief history of gene editing
The discovery of CRISPR-Cas9 has revolutionized the field of gene editing, offering unprecedented precision, efficiency, and versatility compared to earlier technologies. In just a decade since its first use in mammalian cells, CRISPR has transformed biological research and opened up exciting new possibilities for treating genetic diseases.
Gene editing enables gene therapy approaches, where faulty genes causing diseases can be corrected or therapeutic genes can be inserted into cells. Some key applications of gene editing for gene therapy include:
Correcting mutations in cells that cause inherited genetic disorders like sickle cell disease, cystic fibrosis, or muscular dystrophies.
Engineering immune cells like T cells or stem cells to enhance their ability to fight cancers as part of cancer immunotherapies.
Inserting functional genes into cells to compensate for genes that are mutated or missing, allowing production of therapeutic proteins.
Modifying genes involved in other diseases like HIV, liver disorders, blindness, and neurological conditions.
By harnessing gene editing tools like CRISPR-Cas9, scientists can precisely rewrite the genetic code within cells and develop transformative gene therapies for a wide range of diseases with a genetic basis or component.
A brief history on gene editing to get us started:
1960s: Discovery of DNA structure and genetic code laid the foundation.
1970s: First recombinant DNA techniques developed, allowing genes to be isolated and transferred between organisms.
1980s-1990s: Viruses used as vectors to deliver therapeutic genes in early gene therapy trials, with mixed success.
1996: Zinc finger nucleases (ZFNs) developed as first genome editing tool to make targeted DNA cuts.
2000s: Transcription activator-like effector nucleases (TALENs) provided another genome editing approach.
2012: Revolutionary CRISPR/Cas9 system discovered, allowing precise, efficient, and flexible genome editing.2010s: CRISPR/Cas9 rapidly adopted for research, therapeutic development, and first clinical trials.
Present: Continued refinement of CRISPR tools like base editors. Genome editing becoming widely used across biology and medicine.
Let’s look at the recent trends in gene therapy development followed by a science review of CRISPR and other gene editing techniques (if you’re interested in the details!).
📈 Trends & Insights
In recent years, cell and gene therapy treatments have become a major area of research, development, and investment with the notable approval of Vertex Pharmaceuticals’ Casgevy to treat sickle cell disease.
To date, there are 37 FDA approved cell & gene therapy products.
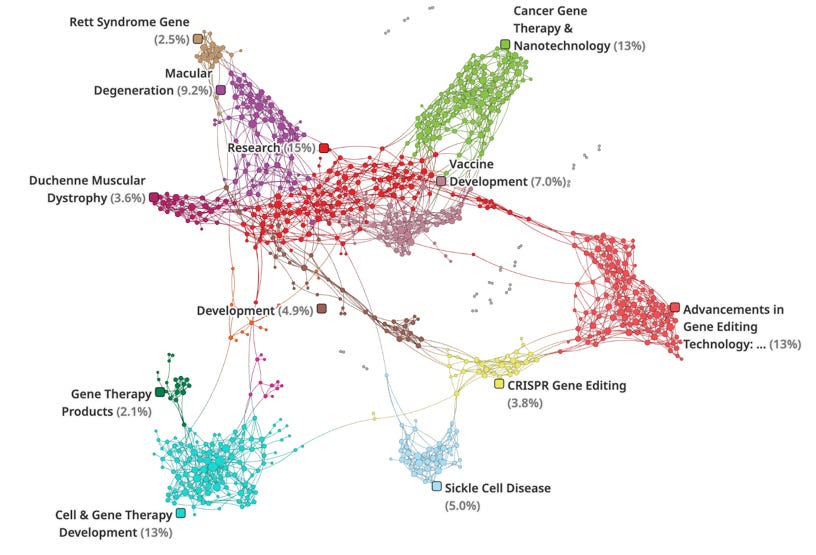
A network analysis of 2023-2024 trends, using open-source data, highlight a strong domain of academic and pharma research (top left, green, purple, mauve, and maroon) with a strong link to the advancements in gene editing technologies (red) and CRISPR gene editing in particular (yellow). This data reflects the rapid adoption and development of CRISPR-based tools for precise genome engineering across various applications.
The lower end of the network highlights the translation of gene editing and cell engineering advances into therapeutic products and clinical development programs. Connections between CRISPR Gene Editing and areas like Sickle Cell Disease (5.0%), Vaccine Development (7.0%), and Rett Syndrome Gene (2.5%), suggesting the application of CRISPR across diverse therapeutic areas.
Cancer Gene Therapy & Nanotechnology (13%) is a prominent area, demonstrating the convergence of gene editing with nanomedicine approaches for developing novel cancer treatments.
Overall, these datapoints indicate a rich focus in research powered by advancements in gene editing that is translating into breakthroughs in therapeutics.
Now, let’s look at the competitive landscape of gene editing companies, cell & gene therapy (C>) companies, and the diseases/applications they are focused on.
Gene Editing and Gene Therapy Landscape
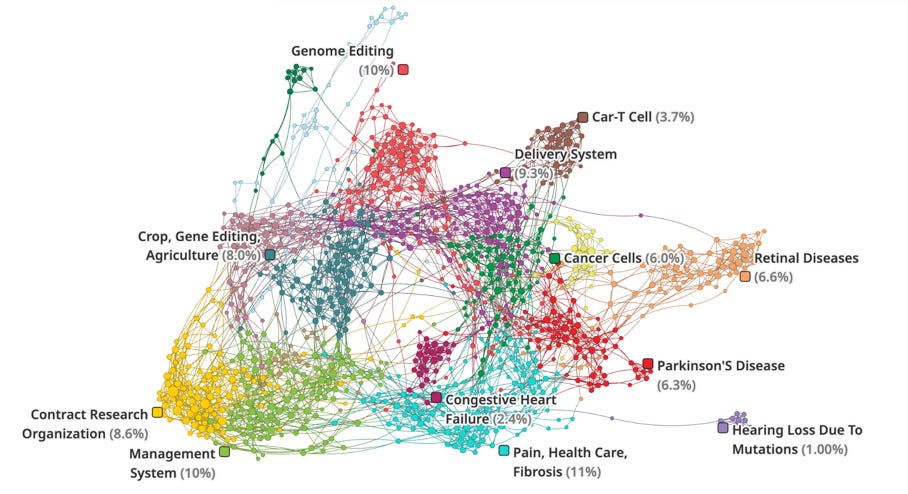
Another network of players in the space include companies working on genome editing, CAR-T cell therapies, delivery systems, cancer treatments, and treatments for genetic disorders like sickle cell disease (e.g. CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, Vertex Pharmaceuticals, Novartis, Gilead, etc.). It’s worth noting that larger pharma companies are partnering with or acquiring gene editing/C> startups to gain capabilities (e.g. Vertex Pharmaceuticals & Exonics Therapeutics, Sanofi & MeiraGTx).
The applications of gene editing span a wide range of diseases like cancer, genetic disorders, vision loss, neurological conditions, and even agricultural applications for crop gene editing.
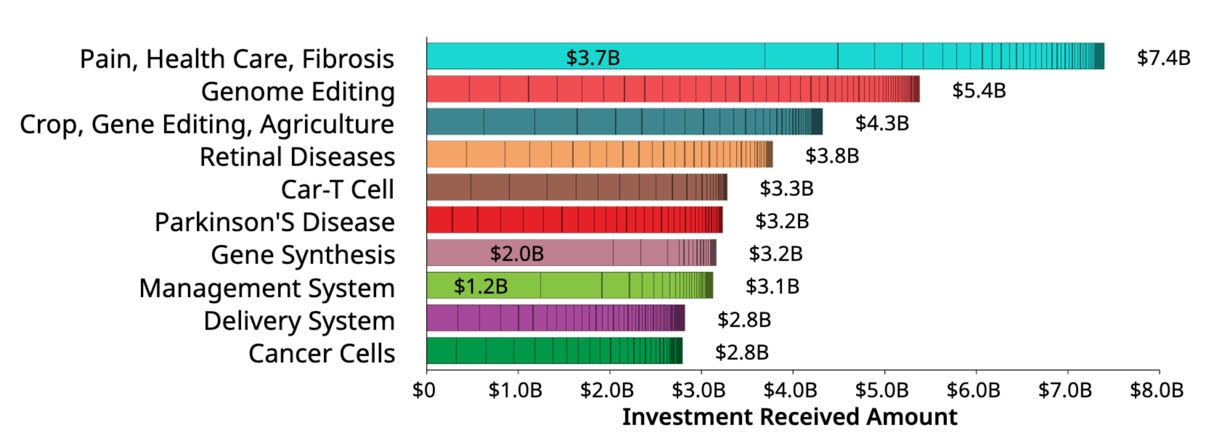
Looking at investment amounts, the bar graph displays the investment received for various areas of gene therapy research and development. It highlights the significant funding being directed towards different applications of gene therapy technology.
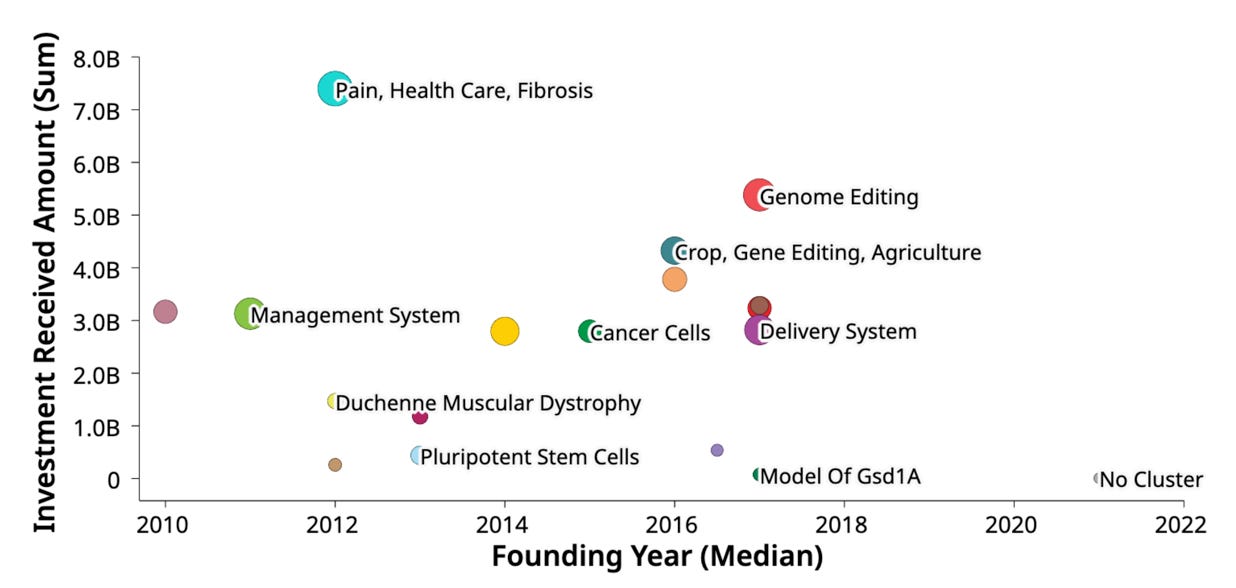
Next, the scatter plot graph shows the investment received (in M $USD) for various research and development areas in the field of biotechnology or life sciences, plotted against the founding year (median) from 2010 to 2022. The graph shows a general trend of increasing investment over time for most research areas, with higher funding levels observed in more recent years (closer to 2022). The distribution of investment amounts and the founding years (median) for different research areas provides insights into the prioritization and growth trajectories of various biotechnology sectors over the past decade
The research areas with the highest investment amounts are "Pain, Health Care, Fibrosis" and "Genome Editing", both receiving over $7 million in funding.
Other areas like "Crop, Gene Editing, Agriculture", "Management System", "Cancer Cells", and "Delivery System" have received moderate levels of investment, ranging from around $3 million to $5 million.
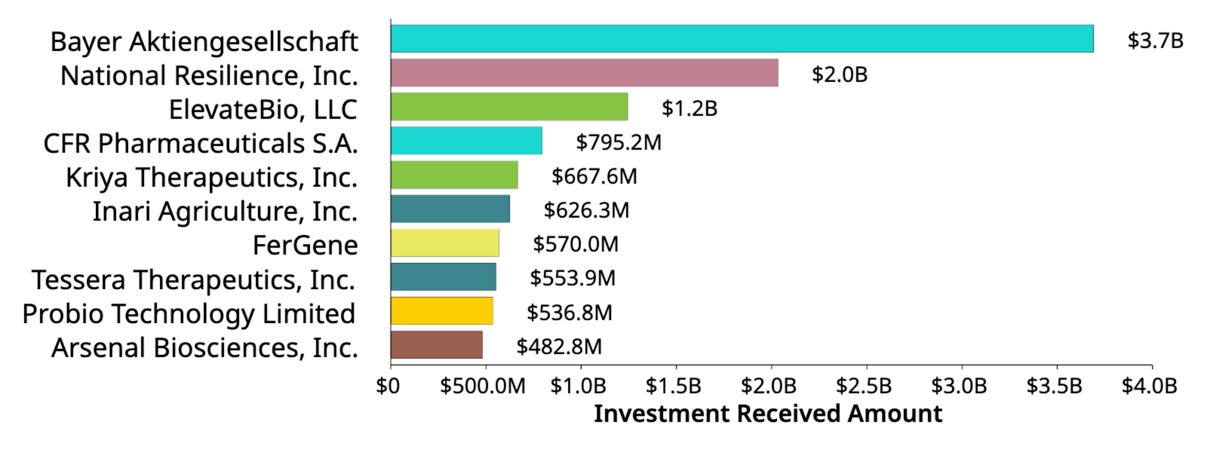
Key companies who have received significant investment in this area as shown below. Top investments are:
Bayer is actively investing in and developing gene therapies by establishing a Cell and Gene Therapy Platform, strengthening its portfolio through collaborations and partnerships, investing in manufacturing facilities, and building an extensive pipeline to address a range of inherited and acquired diseases.
Resilience is a gene therapy manufacturing company that provides end-to-end services to accelerate the development of gene therapies by offering viral vector manufacturing, process and analytical development, innovative platforms like SnapLVV, and GMP manufacturing services and capacity.
ElevateBio is a technology-driven company that powers the development of transformative cell and gene therapies by providing end-to-end capabilities, including gene editing, induced pluripotent stem cells (iPSCs), RNA, cell, protein, and vector engineering, as well as cGMP manufacturing through its BaseCamp facility, to accelerate the design, manufacturing, and development of these innovative therapies.
Outlook and Market Trends
While only 37 cell and gene therapies have been approved by the FDA so far, the pipeline is rapidly growing with over 1,500 ongoing clinical trials, indicating significant future potential.
However, the success rate from Phase 2 to approval for cell and gene therapies is only around 14%, lower than traditional drugs, highlighting development challenges.
Forecasts predict the number of new cell and gene therapy approvals could increase 4-fold in 2024 and 10-fold by 2027 compared to current levels. However, historical approval rates suggest these optimistic forecasts may be tempered.
Orphan drug designation, providing incentives for rare disease treatments, is becoming an important strategic focus for cell and gene therapy companies.
Key challenges include improving delivery methods, reducing off-target effects, increasing editing efficiency, managing costs, and establishing robust manufacturing capabilities.
While still an emerging field with scientific and regulatory hurdles, the gene editing and cell & gene therapy landscape is rapidly evolving, with major investments, a growing pipeline, and significant potential to transform treatment across multiple disease areas in the coming years.
🧪 The Science Review
Let’s dive into the basics of CRISPR, its therapeutic applications, recent breakthroughs, and other techniques.
CRISPR-Cas9: Basics and Mechanism
CRISPR, short for clustered regularly interspaced short palindromic repeats, is a naturally occurring bacterial defense system that scientists have adapted for genome editing. The CRISPR-Cas9 system consists of two key components[4]:
Guide RNA (gRNA): A small piece of RNA with a "guide" sequence that binds to a specific target sequence in the DNA.
Cas9 enzyme: A nuclease that cuts the DNA at the targeted location.
When the guide RNA recognizes the target DNA sequence, it directs the Cas9 enzyme to make a double-stranded break at that precise location. The cell then attempts to repair this break, which researchers can exploit to introduce specific genetic changes.
CRISPR in Gene Therapy
The ability to precisely edit genes has immense potential for treating genetic diseases. CRISPR-based gene therapies aim to correct disease-causing mutations, silence harmful genes, or introduce protective mutations. Some key applications include:
Blood disorders: CRISPR is being used to correct mutations causing sickle cell disease and beta-thalassemia by editing hematopoietic stem cells[3].
Cancer immunotherapy: CRISPR-engineered T cells with enhanced tumor-killing abilities, such as CAR-T cells with PD-1 knocked out, are showing promise in clinical trials[3][9].
Rare genetic diseases: CRISPR has the potential to treat a range of rare disorders by correcting the underlying genetic defects[3][9].
While still in early stages, these CRISPR-based therapies are rapidly advancing towards the clinic. The first CRISPR therapy, Vertex's CTX001 for sickle cell disease and beta-thalassemia, is expected to be approved in 2023.
Key Achievements in CRISPR Field
In its first decade, CRISPR has enabled several groundbreaking achievements:
Rapid disease modeling: CRISPR has accelerated the creation of cellular and animal models of human diseases, facilitating research into underlying mechanisms and drug discovery[5][6].
Base and prime editing: Advanced CRISPR tools like base editors and prime editors enable precise single-base changes or targeted insertions without double-strand breaks, expanding therapeutic possibilities[9][12].
In vivo gene editing: Delivery of CRISPR components directly into the body to correct disease mutations in target organs is becoming feasible, with promising results in animal models[6][12].
Expanded genome targeting: Variants like Cas12a and engineered Cas9 are overcoming limitations of the original system, such as PAM sequence requirements, expanding the range of editable sites[2][12].
Clinical trials: Numerous CRISPR-based therapies have entered clinical trials for diseases like cancer, blood disorders, and inherited blindness, with initial results demonstrating safety and efficacy[6][11].
Recent Trends in CRISPR Research
CRISPR technology continues to evolve at a rapid pace. Some exciting recent developments include:
CRISPR-based diagnostics: CRISPR components are being harnessed to create sensitive diagnostic tests for infectious diseases, cancers, and more.
Epigenetic editing: CRISPR tools are being adapted to modify epigenetic marks, allowing researchers to study and manipulate gene regulation without altering the underlying DNA sequence.
Genome-wide screens: CRISPR enables efficient screening to identify genes involved in specific biological processes or disease pathways, accelerating drug target discovery.
Expanded delivery methods: Novel viral and non-viral CRISPR delivery systems are being developed to improve efficiency, specificity, and safety for clinical applications.
Multiplexed editing: Strategies to perform multiple targeted edits simultaneously are enhancing CRISPR's efficiency and expanding its utility for complex disorders.
As the technology continues to advance and more therapies enter clinical testing, CRISPR is poised to reshape the future of medicine. However, responsible development and thoughtful oversight will be crucial to ensuring that this powerful tool benefits patients while minimizing risks.
Beyond CRISPR: Exploring the Landscape of Non-CRISPR Gene Editing Platforms
The advent of CRISPR-Cas9 technology has revolutionized the field of gene editing, offering unprecedented precision and efficiency in modifying DNA sequences. However, as researchers continue to push the boundaries of genome engineering, they have also been exploring alternative, non-CRISPR based approaches that could overcome some of the limitations of CRISPR and expand the toolbox of gene editing technologies.
ZFNs and TALENs: The Pioneers
Before CRISPR took the world by storm, zinc finger nucleases (ZFNs) and transcription activator-like effector nucleases (TALENs) were the leading tools for gene editing. Both ZFNs and TALENs consist of a DNA-cleaving nuclease fused to a DNA-binding domain, allowing them to target specific DNA sequences.
ZFNs, in particular, have a long history, with Sangamo Therapeutics being the first company to use them for ex vivo and in vivo gene editing. While ZFNs can be difficult and expensive to engineer, they have been used in clinical trials. TALENs, on the other hand, are easier to design than ZFNs and have been used by companies like Cellectis to engineer CAR-T cells. Interestingly, a recent study suggests that TALENs may be more efficient than CRISPR-Cas9 for targeting heterochromatin regions.
ARCUS: Small but Mighty
Developed by Precision BioSciences, ARCUS is a novel gene editing platform that uses endonucleases derived from a genetic element found in algae (I-CreI). What sets ARCUS apart is the small size of its nucleases, which could potentially enable easier delivery to target cells. This feature makes ARCUS an attractive option for in vivo gene editing applications.
Synthetic Mobile Genetic Elements: A Natural Approach
Tessera Therapeutics is taking a unique approach to gene editing by developing synthetic mobile genetic elements (MGEs). Found abundantly in nature, MGEs can insert DNA into genomes without causing double-strand breaks, which are a common byproduct of CRISPR-based editing. By harnessing the power of MGEs, Tessera aims to make diverse genomic alterations without relying on error-prone DNA repair mechanisms.
Others
ADAR Enzymes: Editing RNA instead of DNA: While most gene editing technologies focus on modifying DNA, ADAR (adenosine deaminase acting on RNA) enzymes offer a different approach by editing RNA instead.
Retrons: Enabling High-Throughput Screening: Retrons are another promising non-CRISPR gene editing tool that produce targeted single-stranded DNA, which can generate precise gene editing in vivo through a process called recombineering.
NgAgo and CyDENT: Emerging Alternatives: NgAgo, a prokaryotic Argonaute endonuclease, has garnered attention for its ability to perform gene editing without requiring a PAM motif, which is a limitation of Cas9. While initially controversial, recent studies have demonstrated NgAgo's potential for genetic engineering.
The Future of Gene Editing
As the field of gene editing continues to evolve, it's becoming clear that CRISPR-Cas9, while groundbreaking, is not the only player in the game. Non-CRISPR based platforms like ZFNs, TALENs, ARCUS, synthetic MGEs, ADAR enzymes, retrons, NgAgo, and CyDENT are expanding the possibilities of genome engineering, each with their own unique strengths and potential applications.
By having a diverse array of gene editing tools at their disposal, researchers can choose the best approach for their specific needs, whether it's targeting hard-to-reach genomic regions, enabling high-throughput screening, or minimizing off-target effects. As these technologies continue to advance, we can expect to see new breakthroughs in fields ranging from basic research to therapeutic development.
In conclusion, while CRISPR may have stolen the spotlight in recent years, the landscape of gene editing is vast and diverse. Non-CRISPR based platforms are not only complementing CRISPR-based approaches but also pushing the boundaries of what's possible in genome engineering. As we move forward, it will be exciting to see how these technologies evolve and contribute to our understanding and manipulation of the genome.
📚Good Read of the Month
FDA Approves First Cellular Therapy to Treat Patients with Unresectable or Metastatic Melanoma
A close partner of gene therapy, cell-based therapies have reached a major milestone this year to treat advanced melanoma, providing a new cellular therapy option for patients whose cancer has progressed despite other available therapy.
In March 2024, the U.S. Food and Drug Administration (FDA) approved Amtagvi (lifileucel), the first cellular therapy for treating adult patients with unresectable or metastatic melanoma. This approval is for patients whose cancer has progressed after treatment with other therapies, such as PD-1 blocking antibodies and, if positive for the BRAF V600 mutation, a BRAF inhibitor with or without a MEK inhibitor.
Key points:
Amtagvi represents a novel T cell immunotherapy for patients with limited treatment options for this aggressive form of skin cancer.
Melanoma is often caused by exposure to ultraviolet light and has a significant risk of metastasizing to other parts of the body.
Prior treatments for unresectable or metastatic melanoma include immunotherapy using PD-1 inhibitors and drugs targeting the BRAF gene for melanomas with BRAF mutations.
Amtagvi was approved through the Accelerated Approval pathway, which allows for earlier approval of drugs that treat serious conditions and fill an unmet medical need based on a surrogate endpoint.
As always, feel free to reach out to me via email to discuss how these analysis strategies can support your business.
Cheers,
Mida